Verify mobile number to view the solution
Solutions
Explanation:
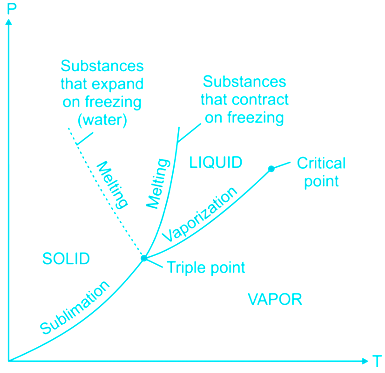
"Extreme points of fusion curve are triple point and critical point" is an incorrect statement
Validation:
From the phase diagram, it is seen that the triple point and critical point are the extreme points of the vaporization curve.
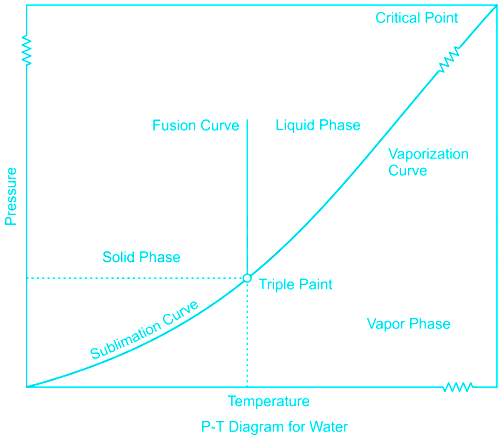
"Extreme points of vaporization curve are triple point and critical point" is a correct statement
Validation:
From the phase diagram, it is seen that vaporization starts from the triple point and goes till critical point beyond which liquid is directly flashed into vapour and there is no vaporization.
"Sublimation curve, fusion curve, and vaporization curve meets at critical point" is an incorrect statement
Validation:
The sublimation curve fusion curve and the vaporization curve meets at the triple point.
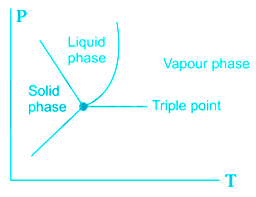
"Fusion curve for water has positive slope" is an incorrect statement
Validation:
The fusion curve of water has a negative slope it can be seen on the phase diagram also, the reason for this negative slope is that water expands after freezing, unlike other substances which contract after freezing.