Verify mobile number to view the solution
Solutions
Explanation:
Work transfer and heat transfer are a path function.
Example:
It is possible to take a system from state 1 to state 2 along many quasi-static paths such as A, B or C. Since the area under each curve represents the work for each process, the amount of work involved in each case is not a function of the end states of the process and it depends on the path which the system follows in going from state 1 to state 2. For this reason, work is called a path function, and δW is an inexact or imperfect differential.
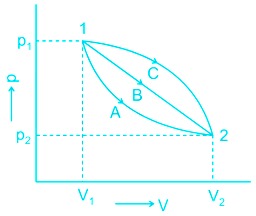
According to the question:
For the given problem statement i.e. “work done in an adiabatic process”, the path is fixed i.e. adiabatic process.
And for a given path work done will be dependent on the end states only.
So, the work done in an adiabatic process between two given end states depends on end states only.
or, from first law of thermodynamics,
δQ = ΔU + δW,
for an adiabatic process δQ = 0.
i.e. 0 = ΔU + δW
⇒ δW = - ΔU = U1 - U2
Since, work done is equal to change in internal energy and internal energy is a point function or state function.
So, the work done in an adiabatic process between two given end states depends on end states only.





