Verify mobile number to view the solution
Solutions
Concept:
The first law of thermodynamics
For a closed system/non-flow system undergoing a process, (1 - 2)
Q1-2 = ΔE + W1-2 …1)
E = Stored energy of a system
This stored energy can be viewed as the sum of microscopic and macroscopic energies.
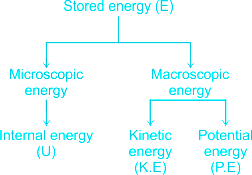
⇒ Q1-2 = Δ (U + KE + PE) + W1-2
⇒ Q1-2 = ΔU + ΔKE + ΔPE + W1-2 …2)
For a non-flow or closed system at equilibrium, ΔKE and ΔPE are negligible,
So, these 2 terms can be neglected.
⇒ Q1-2 = ΔU + W1-2 …3)
Also, for a perfect gas, the internal energy is a function of temperature only.
i.e. dU = mCνdT …4)
Calculation:
Given equation is
Q1-2 = W1-2 …5)
But first law states that; Q1-2 = ΔU + W1-2 …6)
Comparing 5) and 6)
⇒ ΔU = 0 …7)
But for perfect gas; dU = mCνdT, integrating both sides
\(\mathop \smallint \nolimits_1^2 dU = \mathop \smallint \nolimits_1^2 m{C_\nu }dT\)
⇒ U2 – U1 = mCν(T2 – T1) {Assuming constant m, Cν}
ΔU = mCν(ΔT) …8)
Comparing 7) and 8)
⇒ ΔT = 0 {∵ m ≠ 0, Cν ≠ 0}
⇒ T2 = T1 = Constant = Isothermal process
![]()
Remember the properties of perfect gases and apply these directly instead of writing first law.
Study all the basic processes in detail like the adiabatic process, Isobaric, isochoric etc.





