Verify mobile number to view the solution
Solutions
Explanation:
Entropy:
- Entropy is the index of unavailability or degradation of energy. Heat always flows from hot bodies to cold bodies and this becomes degrades or less available.
- For a reversible transfer of heat change in entropy
\({\rm{\Delta }}s = {s_2} - {s_1} = \mathop \smallint \nolimits \frac{{dQ}}{T}\) where, s = entropy, Q = heat, T = temperature
- No process between two equilibrium states is possible if it would result in a decrease in the total entropy of the system and surroundings.
Clausius Inequality:
Clausius inequality states that \(\oint \frac{{{\rm{dQ}}}}{{\rm{T}}} \le 0\)
It provides the criteria for the reversibility of a cycle.
If \(\oint \frac{{{\rm{dQ}}}}{{\rm{T}}} = 0\), the cycle is reversible,
If, \(\oint \frac{{{\rm{dQ}}}}{{\rm{T}}} < 0\), the cycle is irreversible and possible
If \(\oint \frac{{\partial Q}}{T} > 0\), cycle is impossible
![]()
Clausius theorem:
Clausius theorem states that: Any reversible path may be substituted by a reversible isotherm and a reversible adiabatic between the same end states such that the heat transferred during the isothermal process is the same as that transferred during the original process.
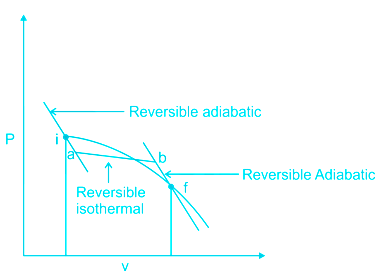
Substitution of a reversible process (i-f) by reversible adiabatic processes (i-a and b-f) and reversible isothermal process (a-b).





