Verify mobile number to view the solution
Solutions
A redox reaction involves the increase in the oxidation state of one species (oxidation) and a decrease in the oxidation state of another species (reduction).
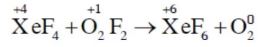
Xenon undergoes oxidation, while oxygen undergoes reduction.





